Eurofins CRL conducts safety and efficacy studies on dental products, such as denture adhesives, tooth-whitening products, mouth washes/rinses, etc. Registered Dental Hygienists and/or licensed Dentists oversee all dental studies.
Examples of studies include: Safety-in-Use studies, which may require a visual examination of the buccal mucosa and tongue before and after the use of the test product; Tooth Whitening Efficacy studies, where the VITA® Shade Guide is used to grade the shade of several teeth before and after the recommended number of product applications; and Denture Adhesive Safety and Efficacy studies, that may require pre- and post-use visual examinations for irritation as well as subjective and/or objective assessment of the force of removal. 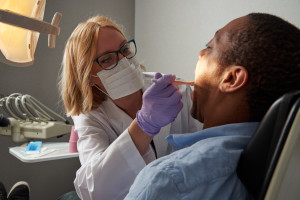
The duration of each study and the number of visits to the clinic will depend on the individual product usage instructions and intended claims for the product. Each study will be designed to meet specific objectives and may include subject questionnaires to obtain consumer opinion of the test product. Eurofins CRL technical staff are available to advise on study design based on the specific product and the claims requiring support.
Individual units of the test product must be provided the Sponsor in a quantity sufficient to last each subject for the required number of uses. Eurofins CRL staff will label each unit with identifying study and subject numbers prior to distribution. Following any required evaluations, subjects are provided with the test products, instructions for use, and a daily diary in which to record each use. At the end of the specified study period, subjects return for a final examination and completion of a questionnaire, if applicable.
Reporting of Results
Following study completion, unaudited topline results may be provided verbally or in writing. All study results will be provided in an audited final report.
Scheduling a Dental Study
To schedule, please contact your Client Services representative at 732-981-1616 or contact us by e-mail: infonj@cpt.eurofinsus.com.
